Imaging Shows Brain Enzyme's Role in Depression
In patients experiencing a major depressive episode, elevated levels of the brain enzyme responsible for breaking down the monoamine neurotransmitters may be responsible for a wide variety of presenting symptoms, a new imaging study suggests.
Researchers at the University of Toronto and Canada's Center for Addiction and Mental Health (CAMH) reported in the November Archives of General Psychiatry that levels of monoamine oxidase A (MAO-A) appear to be significantly elevated throughout multiple brain regions in patients who are in the midst of a major depressive episode and have not taken antidepressant medications.
The increased levels of the enzyme could explain a putative reduction in available monoamines—serotonin, norepinephrine, and dopamine—thought for more than three decades to be at the root of mood disorders.
The team of researchers, led by Jeffrey Meyer, M.D., Ph.D., an associate professor of psychiatry at the University of Toronto and lead researcher in CAMH's Neurochemistry of Depression Program, used positron emission tomography imaging to determine whether brain MAO-A, the enzyme responsible for breaking down serotonin, norepinephrine, and dopamine in preparation for reuptake was higher in those with untreated depression than in healthy control subjects.
The brain-imaging technique showed that in the patients experiencing a major depressive episode, MAO-A was significantly higher in every brain region that the researchers investigated. On average, MAO-A was 34 percent higher in patients with untreated depression than in matched control subjects.
“In major depression,” Meyer said in a press release,“ higher levels of MAO-A is the primary process that lowers monoamine levels. Having more MAO-A leads to greater breakdown of key chemicals like serotonin.”
The report includes a detailed new monoamine model of depression, based on the current imaging study and four previously published studies from Meyer and his collaborators at CAMH.
Meyer emphasized that the lack of a clear and precise disease model has long been a key barrier to making advances in depression treatment.“ Having a disease model is like having a map,” he said.“ Once you have that map, you can really begin to understand how an illness like depression works and offer more targeted and effective treatment.”
Meyer's model of depression (see figure below) hypothesizes that in addition to elevated levels of MAO-A, monoamine transporters also play an important role in removing monoamines from active binding sites across the synapse. Elevated levels of a particular transporter may aggravate already lowered levels of a monoamine due to increased Mao-a by increasing reuptake.
“An important aspect of our advanced monoamine model is that individuals with depression lose chemicals like serotonin and dopamine at different rates based upon transporter density,” Meyer explained.“ This helps explain why one person with depression may experience [certain symptoms] while another may not, and [why] some people have more severe symptoms than others.”
The next step for researchers, Meyer and his team said, will be to investigate why MAO-A levels are elevated in the brain of people with depression. With this understanding, it might be possible to develop targeted preventive therapies as well as more specific therapies for acute treatment.
“Elevated Monoamine Oxidase A Levels in the Brain: An Explanation for the Monoamine Imbalance of Major Depression” is posted at<http://archpsyc.ama-assn.org/cgi/content/abstract/63/11/1209>.▪
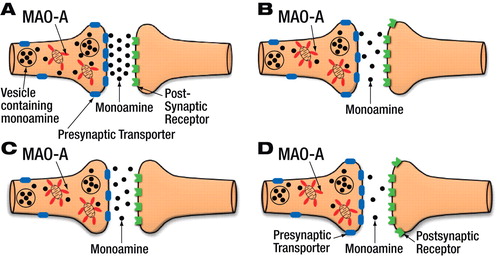
PET imaging data suggest increased activity of monoamine oxidase A (MAO-A) during a major depressive episode (B), resulting in reduced monoamine activity in the synapse, compared with healthy subjects (A). This elevation, paired with fewer monoamine transporters during a mood episode, only modestly reduces available monoamine and leads to mild-to-moderate symptoms (C). However, if increased MAO-A activity occurs with increased numbers of transporters, a severe depletion in available monoamines could occur, possibly leading to more severe symptoms (D).



